Understanding FDA Labeling Requirements Medical Devices
21st Jan 2026
Key Highlights
-
Labels for medical devices sold in Canada must be in both English and French.
-
Labeling requirements vary based on the device's risk class, with higher-risk devices needing more detailed information.
-
Every label must include the device name, manufacturer details, a control number, and intended use.
-
Accurate labeling is crucial for the safe and effective use of the device by both health care professionals and the general public.
-
Failing to meet these labeling requirements can lead to serious regulatory actions, including fines and product recalls.
When you see a medical device, how often do you think about its label? This label is more than just a name tag; it’s a critical communication tool.
Proper medical device labeling acts as the bridge between the manufacturer, health care providers, and you, the user. It ensures everyone understands how to use the device safely and effectively.
Protecting public health is the primary goal, and clear, accurate labeling is the first step in achieving that objective.
Why Accurate Medical Device Labelling Matters?
Accurate medical device labelling isn’t just a regulatory checkbox, it plays a critical role in keeping patients safe and ensuring healthcare teams can work confidently.
Labels are often the first and most important source of information for anyone using a device, from surgeons to lab technicians to everyday consumers.
Why accurate labelling matters under Health Canada:
-
Clarifies the device’s intended purpose, reducing the risk of misuse across both clinical and consumer environments.
-
Delivers critical bilingual safety warnings, helping prevent harm, especially for higher-risk Class II, III, and IV devices.
-
Guides proper installation, handling, and storage, supporting reliable performance throughout the device’s lifecycle.
-
Empowers healthcare professionals to make safe, informed decisions, backed by precise operational and safety information.
-
Enables full traceability through identifiers like lot or serial numbers, which are essential for recalls and incident investigations.
-
Ensures compliance with federal regulatory requirements, protecting manufacturers from penalties, license complications, or product withdrawal.
-
Builds trust with healthcare providers and patients, demonstrating a commitment to safety, quality, and transparency.
When labelling is precise, compliant, and built to withstand real-world conditions, everyone benefits, from manufacturers to clinicians and, ultimately, the patients whose care depends on it.
What Are Health Canada's Labelling Requirements for Medical Devices?
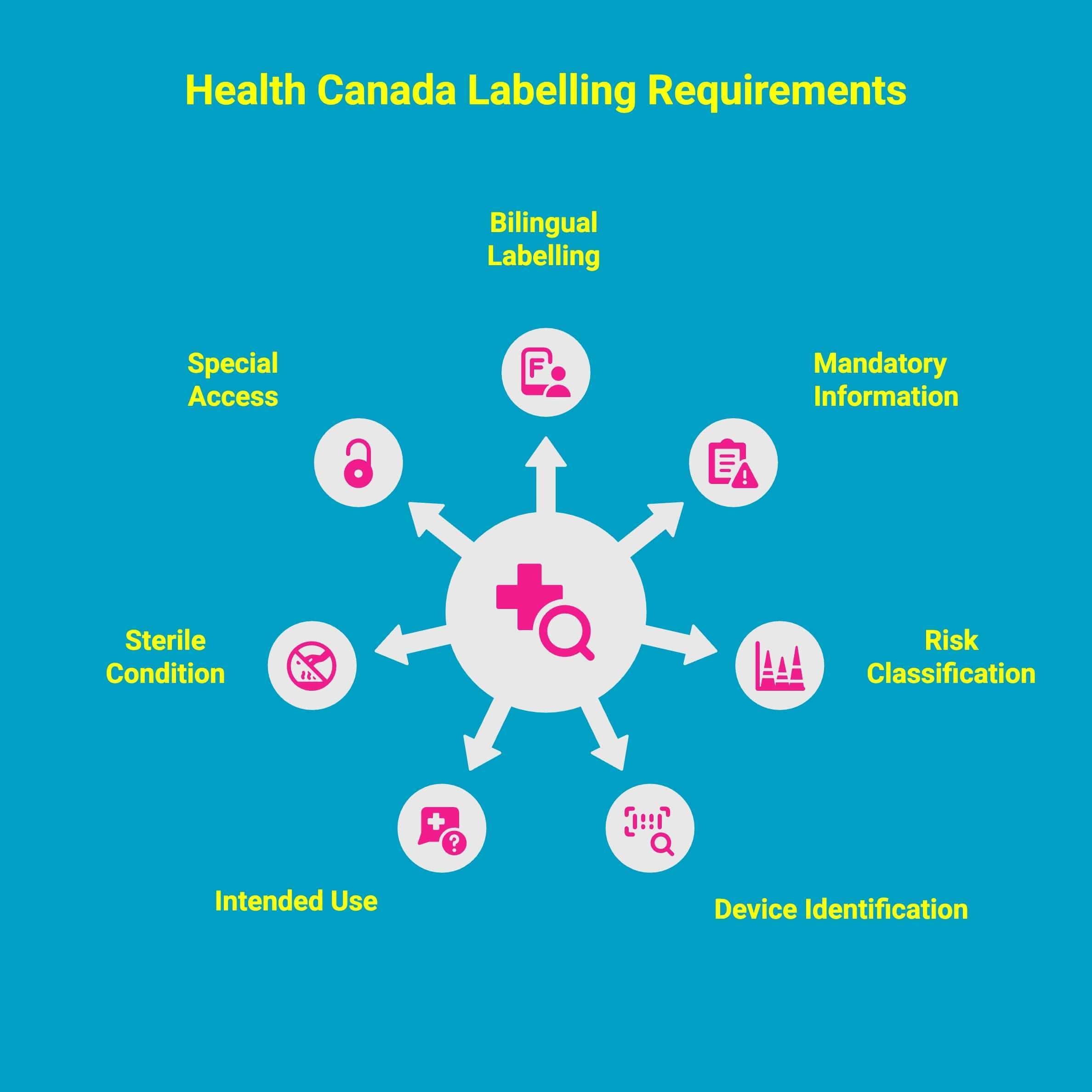
Health Canada sets clear labeling expectations to ensure every medical device is understood, used safely, and properly identified. Canada classifies devices from Class I (lowest risk) to Class IV (highest risk), with labeling requirements increasing as device risk increases.
In the sections below, we explore these requirements in depth, so you know exactly what’s needed for full Health Canada compliance.
1. Bilingual Labelling At The Time Of Sale
All medical device labels in Canada must present required information in both English and French at the time of sale. This includes core safety details, instructions, and identification elements so users across the country can understand the device without confusion.
2. Mandatory Information And Applicable Requirements
Every label must follow the applicable requirements outlined in the Medical Devices Regulations. This includes listing the name of the manufacturer, the unique lot or control number, and the name of the device so it can be accurately tracked throughout its lifecycle.
3. Risk Classification And Higher Risk Device Expectations
Labelling rules vary depending on the device’s risk class. While Class I devices have fewer expectations, higher risk categories must provide more extensive information, including warnings, precautions, and clarity on operational limitations to support safe and consistent use.
4. Device Identification And Name Of The Device
Each device must be clearly identified on the label. This includes the name of the device, the manufacturer’s information, and traceability data that allows regulators and users to confirm compliance and authenticity quickly.
5. Description Of The Medical Condition And Intended Use
Labels must include a clear description of the medical condition the device is designed to diagnose, manage, or treat. This ensures users understand the intended use and helps avoid misuse or application beyond the device’s tested scope.
6. Sterile Condition, Useful Life, And Projected Useful Life
For devices that need to remain in a sterile condition, the label must specify this along with clear handling instructions. Manufacturers must also state the useful life or projected useful life of the device to guide replacement schedules and prevent safety issues.
7. Special Access And Additional Instructions For Complex Devices
Some devices fall under special access pathways, and their labels must reflect any unique requirements tied to that process. More complex products also require detailed instructions and warnings to ensure that health professionals and patients can use them safely and effectively.
Durafast printers make it easier for manufacturers to stay compliant by producing clear, high-contrast labels and supporting efficient dual-language templates that align smoothly with Health Canada’s labelling expectations and regulatory requirements.
What Information Is Required on a Medication Label and How Printing Technology Helps?
It is important to understand what information needs to be on a medication label for label makers and manufacturers alike. Here is a breakdown of the essential information you should find on a device label:
|
Information Category |
Details Required |
|---|---|
|
Identification |
Device name, model number, catalogue number, or unique identifier |
|
Manufacturer Information |
Full name and address of the manufacturer or authorized representative |
|
Traceability |
Lot number, batch number, or serial number for recall and tracking |
|
Expiry Information |
Use-by date or expiration date, when applicable |
|
Directions for Use |
Instructions for operation, storage conditions, and essential warnings |
When these details are clearly presented and durable enough to withstand real-world medical environments, they help protect patient safety, streamline clinical workflows, and ensure every device can be confidently tracked and used exactly as intended.
What Are Some Common Labelling Challenges in Medical Environments?
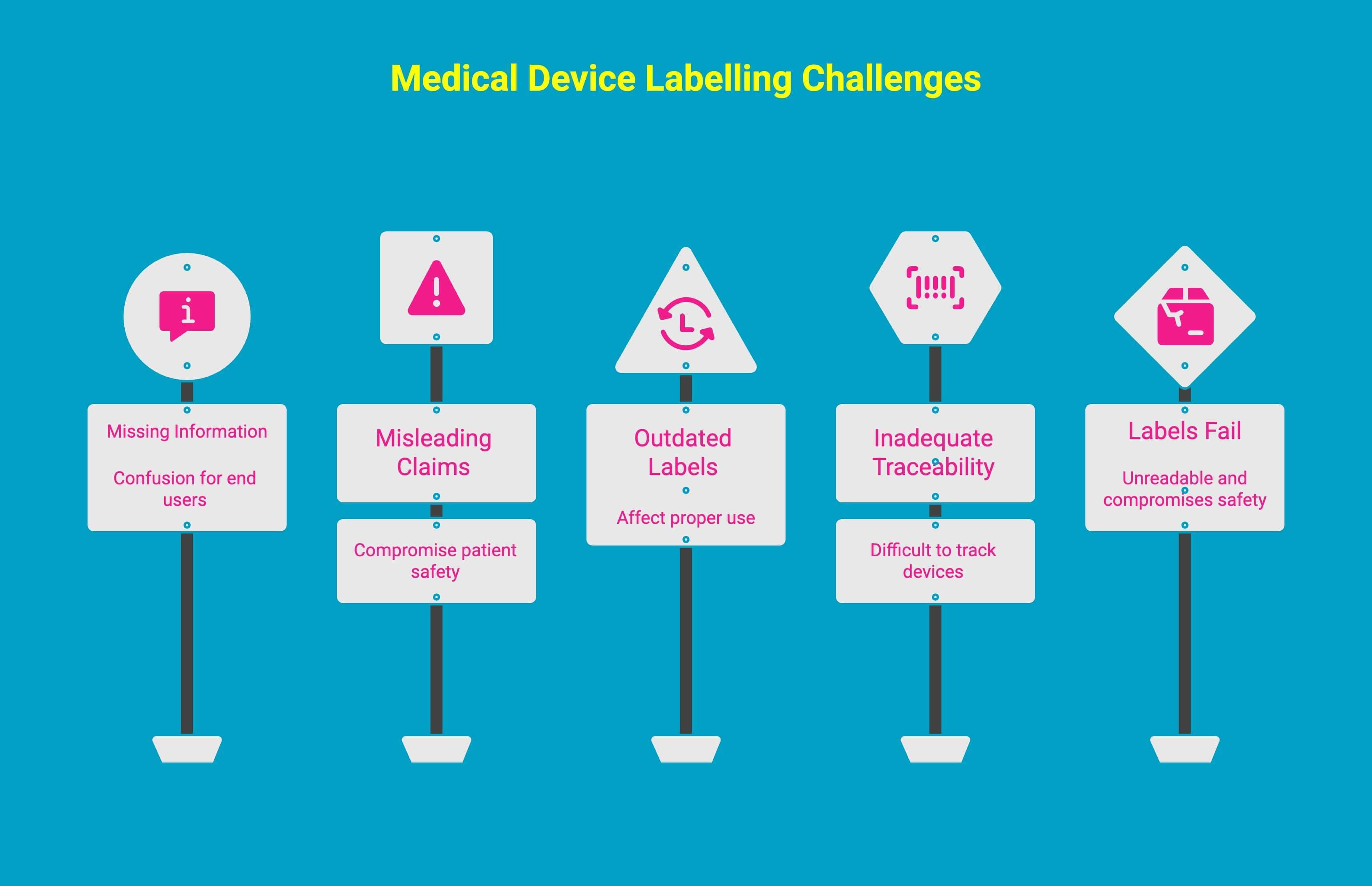
Medical device manufacturers often face recurring labelling challenges as they navigate strict regulatory expectations. Problems such as incomplete information, unclear translations, outdated instructions, and labels that can’t withstand medical environments can quickly lead to safety risks and compliance violations.
1. Missing Or Incorrect Information
One of the most common issues is failing to include mandatory details, such as bilingual text, accurate warnings, or proper usage instructions.
Missing French translations or incorrect terminology can result in confusion for end users and immediate non-compliance.
2. Unapproved Or Misleading Claims
Another challenge arises when labels make claims that go beyond what has been cleared or licensed by regulatory authorities.
Suggesting an unapproved intended use, overstating benefits, or omitting necessary limitations can mislead users and compromise patient safety.
3. Outdated Labels After Design Changes
Device designs, materials, and components are often updated over time, but labels don’t always get revised accordingly.
When a label doesn’t reflect the current design, instructions, or performance data, it creates discrepancies that can affect proper use and regulatory alignment.
4. Inadequate Traceability And Identification
Medical environments rely heavily on traceability. Missing lot numbers, serial numbers, manufacturing dates, or distributor details can make it difficult to track devices, manage recalls, verify authenticity, or conduct quality investigations.
5. Labels That Fail Under Medical Environmental Conditions
Labels used in clinical or laboratory settings must withstand sterilization cycles, moisture, refrigeration, and chemical exposure. If a label peels, smears, or fades during routine handling, it becomes unreadable and compromises both safety and regulatory compliance.
Each medical device group must be labelled in a way that supports the proper use of every component, especially when the products are part of a system, and the information must clearly state the intended use of the devices.
What Are The Best Printers for Medical Device Labelling Requirements?
Choosing the right printing technology is crucial for meeting the stringent requirements for a medical device label. You need printing solutions that produce clear, durable, and compliant labels every time. The best printers are those that can handle the specific materials required to withstand harsh environmental conditions, such as sterilization, refrigeration, and exposure to chemicals, without compromising the integrity of the label information.
The Seiko SLP-720RT, SLP-721RT, and SLP-700 Series are designed to handle the precision, durability, and consistency required in medical environments.
1. Seiko SLP-720RT
The SLP-720RT is built for fast, reliable performance, making it an excellent choice for facilities that require high-volume, compliant label production.
For medical device labeling in Canada, official regulations can be found on the Health Canada website. These documents detail requirements for compliant label production to ensure labels meet Canadian standards.
Key Features:
-
High-resolution printing for razor-sharp barcodes, symbols, and micro-text, making it easy to identify surgical tools and equipment even after multiple sterilization rounds.
-
Works with durable label materials that survive freezing and autoclave heat, perfect for lab vials stored in deep freezers or trays going through steam sterilizers.
-
Fast print speeds that keep workflows moving, especially helpful during peak hospital admissions when wristbands and sample labels need to be created quickly.
-
Simple, quick-load design that reduces downtime and avoids labeling mistakes, giving staff more time to focus on patient care, not printer issues.
2. Seiko SLP-721RT
The SLP-721RT is engineered for environments that demand both precision and long-lasting label durability, especially where chemical exposure is common.
Key Features:
-
Enhanced print clarity for regulatory-level precision, ideal for serialized implant tracking labels that must be perfectly readable every time.
-
Compatible with strong synthetic and polyester label stock, great for labeling diagnostic machines, cables, and tools that are constantly wiped down with disinfectants.
-
Excellent resistance to chemicals, moisture, and fading, ensuring sterilization logs and equipment tags don’t smear or disappear over time.
-
Perfect for high-accuracy, small-batch jobs, such as preparing specialty surgical kits or tracking limited-run medical device components.
3. Seiko SLP-700 Series
The SLP-700 Series provides versatility and consistency, making it a dependable option for medical manufacturers with diverse labelling needs. This reliability is especially important in light of recent updates to Health Canada’s medical device labeling regulations, which emphasize clear, durable labeling and traceability.
Key Features:
-
Handles a wide range of medical-grade label materials, from catheter packaging to medication tray identifiers and safety instruction labels.
-
Reliable, consistent print output that supports traceability and compliance, making barcode scanning smooth and error-free throughout the supply chain.
-
Compact design that fits comfortably in labs, cleanrooms, and assembly lines, where every inch of workspace matters.
-
Built for everyday accuracy and durability, helping prevent costly errors like mislabelled specimen cups or incorrect package identification.
Medical Device Label Printing in Canada: Why Buy Local with DuraFast Label Company?
At DuraFast Label Company, we make medical device labelling clear, compliant, and efficient. Our Seiko SLP-720RT, SLP-721RT, and SLP-700 Series printers are designed to produce sharp, durable, bilingual labels that meet Health Canada’s strict requirements with ease.
Whether you’re preparing labels for sterilized instruments, diagnostic kits, device packaging, or traceability workflows, our solutions give you the flexibility to print exactly what you need, on demand. With professional print quality, reliable performance, and materials engineered for medical environments, you can streamline compliance while keeping your production process running smoothly.
Ready to upgrade your medical device labelling? Explore our Medical Device Label Printers at DuraFast Label Company today.
Conclusion
Accurate labels not only convey essential information but also enhance patient safety and streamline regulatory processes. By being aware of the specific requirements set forth by various regulatory bodies, manufacturers can avoid common labeling challenges and foster trust with both healthcare professionals and patients. As you handle the complexities of medical device labeling, remember that having the right tools and resources at your disposal can make all the difference.
Frequently Asked Questions
What is the difference between FDA, Health Canada, and EU MDR medical device labeling rules?
All three require core details, but each has unique standards. FDA focuses on intended use clarity, Health Canada mandates bilingual content, and EU MDR imposes the most extensive requirements, particularly for higher-risk devices.
Are there recent updates to FDA medical device labeling regulations?
Yes, updates occur regularly. FDA revisions often refine intended use, warnings, formatting, or readability. Manufacturers should monitor official FDA channels to stay compliant with any new medical device label expectations.
How can I ensure my device labels meet all FDA requirements?
Review your labels against the latest FDA rules, confirm clarity for the intended user, and validate accuracy. Using regulatory checklists and label validation testing helps ensure full compliance with FDA requirements.
What are Class 1, 2, and 3 medical devices classified by the FDA?
The FDA uses a three-tiered classification system based on risk. Class 1 devices are lowest risk, Class 2 moderate risk with special controls, and Class 3 highest risk, supporting or sustaining life. Regulatory requirements increase with risk level.
What are the FDA label changes for 2025?
No specific 2025 changes are confirmed as of now. Manufacturers should monitor FDA announcements for new guidance affecting labeling, such as updates to intended use, warnings, expiration information and more.
What is section 34 of the Canadian medical device regulations?
Section 34 outlines specific Health Canada requirements related to device labeling and documentation, making sure all the information is accurate, safe, and compliant with the respective authority, for the Canadian population.
Does Canada require a CE mark for medical devices?
No. Canada does not use the CE mark; it’s an EU requirement. Devices sold in Canada must meet Health Canada’s Medical Devices Regulations and may require a medical device licence and establishment licence.
What is the best basic label printer?
The best basic label printer delivers crisp, durable text and supports materials used for medical device labels, like the Seiko SLP-700 series. Choose a model that ensures long-term legibility and consistent regulatory-ready output.
Are there specific labeling requirements for in vitro diagnostic devices under Health Canada regulations?
Yes, Health Canada has specific labeling requirements for vitro diagnostic devices. The guidance document outlines details that must appear in both official language versions and clearly describe intended use, patient population, warnings, and traceability information.
